FDA’s quality maturity model explained
The FDA's quality maturity model is a comprehensive framework designed to propel organizations in the life sciences industry towards superior quality practices. This model underscores the importance of not just adherence to regulations, but the instilling of quality principles into all facets of an organization's operations.
The quality management maturity model by FDA is structured on five distinct levels of maturity, each characterized by growing levels of sophistication in quality management. These levels range from an initial phase of 'ad hoc' (Level 1), where the processes are typically uncontrolled and reactive, to the ultimate stage of 'optimized' (Level 5), where continuous process improvement is driven by quantitative feedback and benchmarking against industry standards.
In the 'defined' stage (Level 3), the organization’s processes are characterized by standardization and documentation. Beyond this, the 'managed' stage (Level 4) ensures that processes are measured, controlled, and their quality is predictable. This granular approach allows organizations to assess their current standing and identify areas of improvement more precisely.
Adopting this model facilitates a continuous cycle of process improvement, enabling organizations to be proactive rather than reactive in addressing quality issues. Consequently, it enhances not only regulatory compliance, but also customer satisfaction, employee engagement and overall organizational performance.
Key components of the model
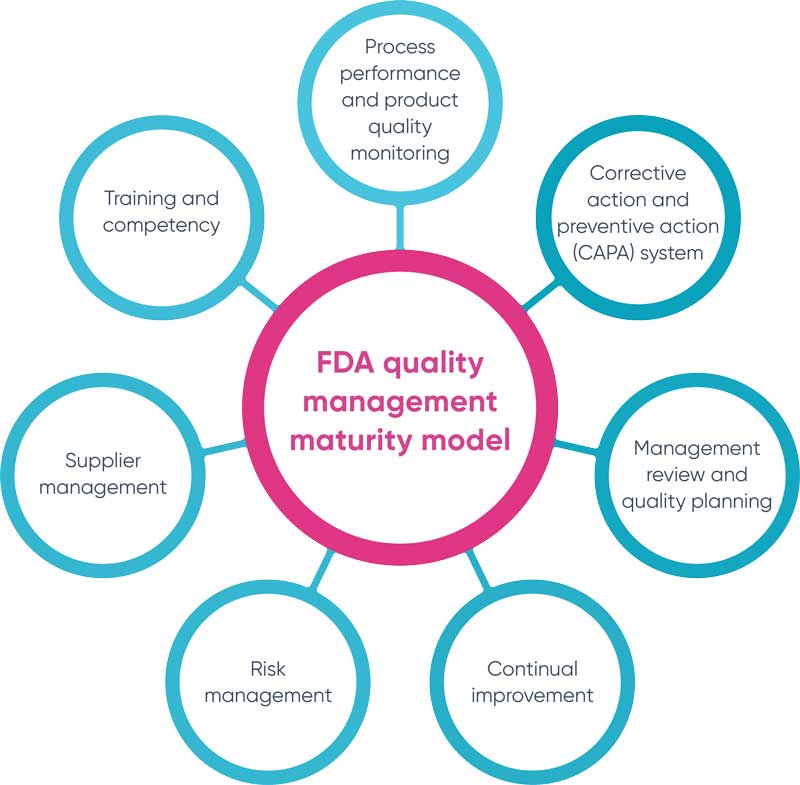
The model is built around seven key areas:
- Process performance and product quality monitoring: Establishing efficient monitoring processes are paramount to controlling and sustaining product quality. This entails adopting a metrics-driven approach to track performance and ensure product consistency.
- Corrective action and preventive action (CAPA) system: This concept focuses on the organization's proficiency in identifying, assessing, and resolving issues as they arise, as well as averting potential problems before they become reality. A comprehensive CAPA system requires an in-depth investigation of root causes, effective corrective and preventive actions and consistent follow-ups to ensure sustained improvements.
- Management review and quality planning: The role of top management is critical in setting quality objectives, reviewing performance against targets and planning for ongoing improvement. Aligning quality practices with overall business objectives ensures appropriate resource allocation.
- Continual improvement: Quality maturity is an ongoing process, with organizations needing to continually identify opportunities for enhancement across all operational areas. This stresses the importance of fostering a culture of continuous learning and innovation to drive quality excellence.
- Risk management: The handling of varying degrees of risk that can impact product quality is inherent to any organization's activities. The FDA model necessitates having a robust risk management system in place to identify, assess and mitigate any potential risks to product quality.
- Supplier management: An organization must ensure that its suppliers meet the same high standards for quality as they do. This involves establishing clear expectations, conducting rigorous audits and continually monitoring supplier performance.
- Training and competency: Investing in employee development is fundamental to achieving consistently high-quality outcomes. This highlights the necessity of ensuring employees are adequately trained and possess the skills required to effectively perform their roles.
By focusing on these seven key areas, organizations can enhance their quality maturity, leading to improved operational efficiency, increased customer satisfaction and a competitive advantage in the marketplace.
The role of Ideagen in your quality maturity journey
Ideagen provide powerful quality management software that serves as an invaluable tool in navigating towards maturity as defined by the FDA's quality maturity model. It offers an integrated platform that allows businesses to manage their operations in accordance with the seven key areas of the FDA's model.
From tracking performance and product quality to managing risks and suppliers, Ideagen provides organizations with the tools they need to align with the FDA's model. It supports the implementation of a robust CAPA system, enables top management review and quality planning and facilitates continual improvement. Additionally, it helps to ensure that employees are adequately trained and competent to carry out their roles effectively.
In essence, Ideagen’s solutions go beyond being a mere tool. We become a strategic partner, enabling organizations to elevate their quality practices, boost operational efficiency and secure a competitive edge.
Aaron is an experienced content writer specializing in quality and compliance, with a focus on regulatory standards and enhancing operational practices. With a strong background in the life sciences and manufacturing industries, he excels at creating clear, impactful content that supports organizations in achieving sustainable excellence and meeting complex industry requirements.


