Whether you manage 5 sites or 500, we've seen your quality challenges before. This demo shows exactly how we solve them – from CAPA to Mazlan AI.
Quality management system. Less admin. More actual quality
One system. Every quality process connected. AI that finds risk before you do. Trusted by 18,500+ organizations.
Trusted by quality teams where failure isn't an option.
All your quality processes. One system. Zero gaps
Non-conformances slip. Documents go stale. Audits become fire drills. Ideagen connects document control, CAPA management, audits, training, supplier quality and inspection in one platform.
Every record traceable. Every workflow structured. ISO 9001, ISO 13485, 21 CFR Part 11, AS9100, IATF 16949 – built in. No bolt-ons.
A QMS built for teams who can't afford to get it wrong
Every standard met. Every record audit ready. Ideagen's quality management system is built so regulated industries never have to chase compliance again.
Healthcare & Pathology
Nuffield Health. South Tees NHS. Both chose our QMS to centralize pathology compliance, streamline CAPA and hit ISO 9001 and ISO 15189 – across every site.
Pharmaceuticals & Manufacturing
Centralized CAPA, calibration, training and EU GMP compliance across 3 sites. Torbay Pharmaceuticals did it. So can you.
Medical Devices & Life Sciences
BSL-3 labs and clinical trials can't run on paper. Duke University Medical Center digitized SOPs, audits and incident reporting – and eliminated the risk.
General Manufacturing
ISO 9001, non-conformance trending and PDCA – built for manufacturing operations. 6,000+ customers use it daily. The results speak for themselves.
The numbers behind the QMS that sets the standard
Core quality management system capabilities

Change Management
Automatically track ISO standards and regulatory changes across all jurisdictions. New requirements embed directly into existing workflows – with full audit trails and clear change impact visibility.

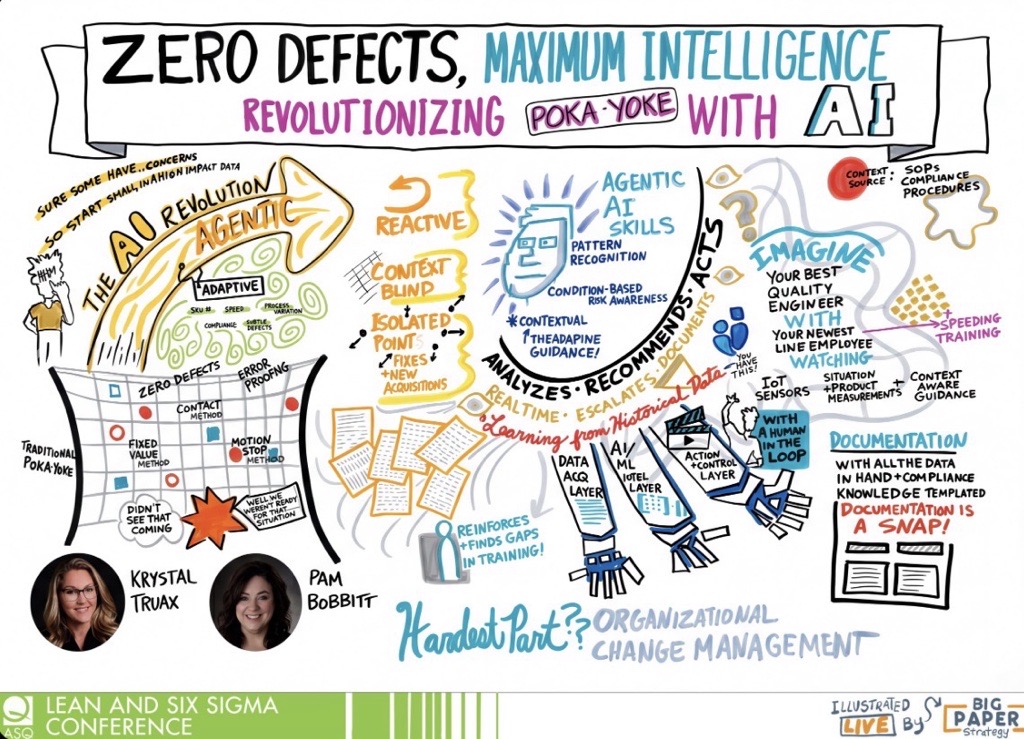
CAPA management
Don't just fix the problem. Stop it coming back. Automated CAPA workflows deliver systematic root cause analysis and predictive insights. Audit scores improve by 25–40%.

Audit management
Audits shouldn't be fire drills. Automated evidence, adaptive checklists and real-time collaboration keep you ready – ISO 15189, MHRA, FDA. Every finding traced to action.

Training and competency management
Your quality standards are only as good as your team's training. Automated competency tracking, refresher scheduling and GCP/ISO alignment – new hire training cut by 40%.

Named a Leader in quality management software
Verdantix's independent Green Quadrant analysis evaluates 14 QMS platforms serving life sciences, healthcare and manufacturing. See how Ideagen earned the highest scores for AI operations, workflow automation and supplier management. Access this $3,000 report courtesy of Ideagen.

AI embedded in regulated quality work. Governed. Auditable
Mazlan doesn't just tell you what happened. It tells you where risk is forming before the audit, before the recall, before the board asks. Three days of investigation becomes three hours. 80%+ adoption on the floor.
Human-approved, every time.
Quality, safety and compliance. Connected in one platform
Your quality management system doesn't work in isolation. Ideagen Hub connects quality, EHS, compliance and risk – same data, same audit trail, same view.
See Ideagen's quality management system in your environment
Explore our quality management system at your own pace
No login. No sales call. No pressure. Just real document control, CAPA and audit workflows running in environments like yours. See it when you're ready.
Frequently Asked Questions
Document control, CAPA management, audit management, inspection management, training and competency management, supplier quality management and regulatory intelligence, all in one connected QMS platform.
SO 9001, ISO 13485, 21 CFR Part 11, AS9100, IATF 16949, ISO 17025 and more built directly into the workflows. You don't check for compliance. You just are compliant.
30 days to go live. Our AI configures itself through natural conversation, not months of requirements workshops. We've done this 18,500 times. We know what you need before you ask.
Every Mazlan AI output – from root cause suggestions to drafted reports – requires human review and approval. Accountability stays with your team. AI does the heavy lifting; you make the decisions.
Yes. Ideagen is built for enterprise-scale quality management. Standardize processes globally while giving regional teams the flexibility they need. One platform. Every site. Full visibility.
Still have questions?
Our team knows your industry. If your question isn't answered above, talk to us. No sales pitch, just answers.