Building an ICH Q10 pharmaceutical quality system with Ideagen quality solutions
An ICH Q10 pharmaceutical quality system serves as a robust framework to seamlessly integrate ISO and GMP quality benchmarks throughout the entire lifecycle of your pharmaceutical products. This comprehensive system ensures that every stage, from initial development to final manufacturing and distribution, adheres to the highest standards of quality and compliance. By adopting ICH Q10, you can systematically manage risks and drive continuous improvement in your processes, ensuring that your products consistently meet the rigorous expectations of both regulatory authorities and your customers.
Additionally, implementing such a system enhances your ability to respond swiftly to any quality issues, thereby maintaining the integrity and safety of your pharmaceuticals. If your operation aims to consistently deliver high-quality pharmaceuticals that satisfy both your customers and regulatory authorities, building an effective and resilient ICH Q10 system is a crucial step towards achieving this goal and sustaining long-term success in the competitive pharmaceutical industry.
To support this endeavor, Ideagen’s integrated quality management software system offers a comprehensive suite of interacting functionalities designed to help you build, maintain and embed ICH Q10 within your organization. This software not only streamlines processes but also enhances traceability, reduces risks and ensures continuous improvement. Read on to learn how.
Effective ICH Q10 operation
An effective quality management system (QMS) is vital in the pharmaceutical world.
ICH Q10 was developed by the International Council of Harmonization to provide an integrated pharmaceutical quality management model, going beyond the benchmarks of Good Manufacturing Practice (GMP) to lay out specific management responsibilities and quality system ingredients in the style of an ISO 9000 framework.
An effective ICH Q10 system gives your pharmaceutical business three operational abilities: to realize your products and bring them to market, to control the end-to-end lifecycle of your products and to drive continuous pharmaceutical quality improvement.
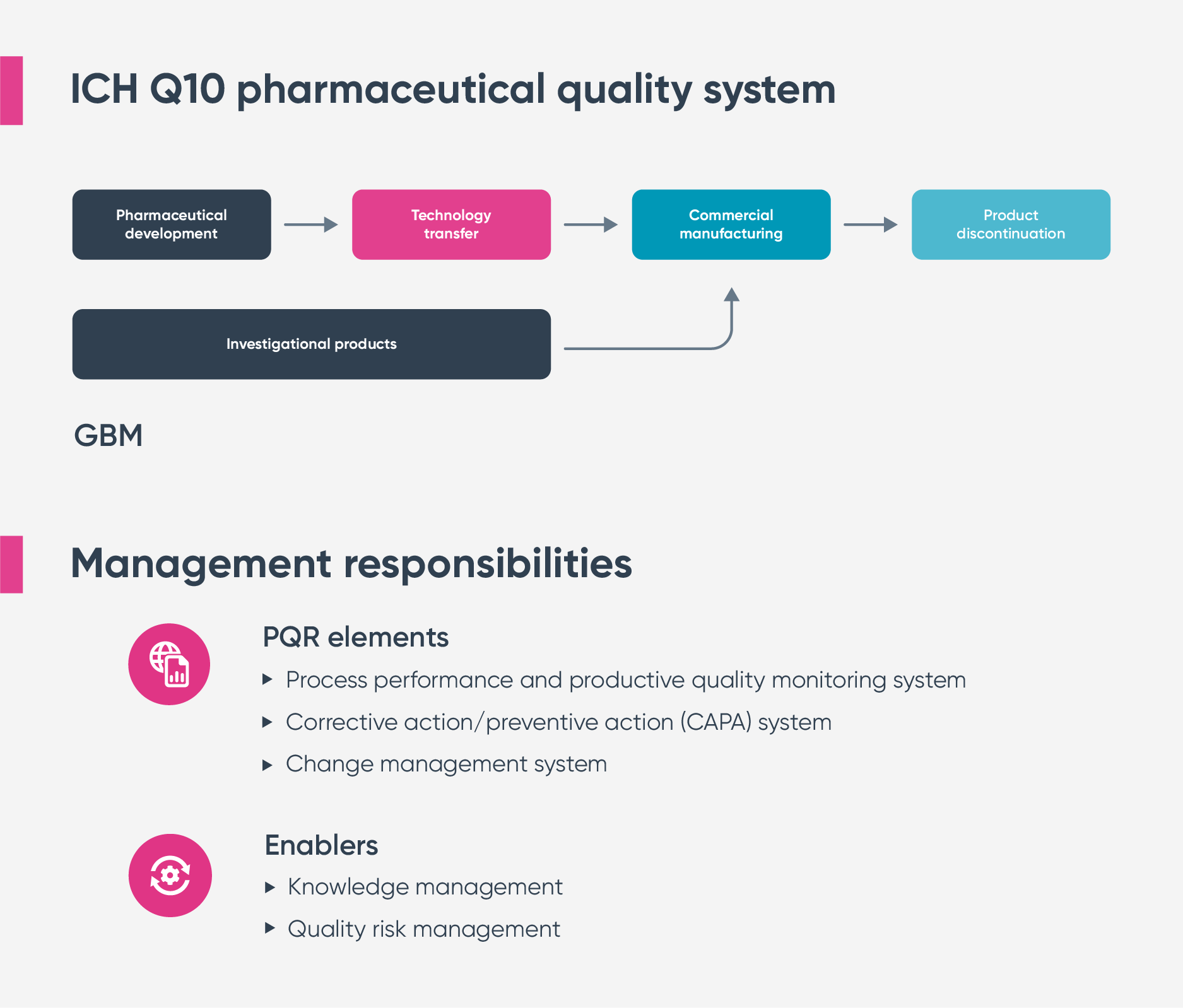
As you can see from the diagram above, ICH Q10 covers four key areas of the product lifecycle:
- Development
The processes and procedures governing how products are ideated and developed, including exploratory and clinical development studies.
- Technology transfer
The transfer of product and process knowledge between development and manufacturing and within or between your operational sites.
- Commercial manufacturing
Controlling and continually improving the product realization and distribution stage
- Discontinuation
Managing the end of the pharmaceutical product lifecycle in a controlled, consistent way and it mandates four core “pillars” or quality system elements to support these areas:
- Process performance and product quality monitoring
- Corrective and preventive action (CAPA) management
- Change management
- Management review
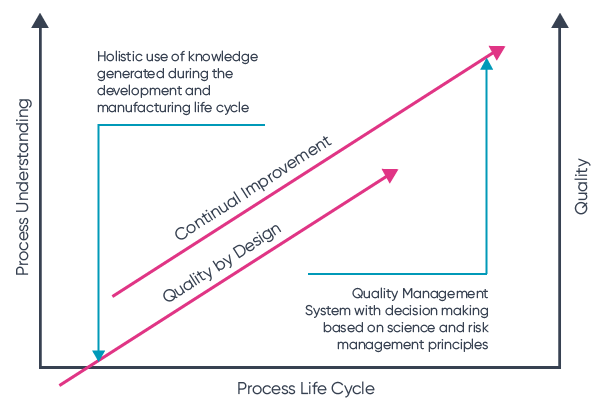
The role of an electronic quality management system
Ideagen Quality Management is designed to provide regulated businesses with the holistic, integrated digital toolkit you need to ease compliance burden and embed natural, automatic, day-to-day quality improvement.
Let’s take a look at how each pillar of your ICH Q10 system can be strengthened, streamlined and automated with our quality management software.
Process performance and product quality monitoring
This element of your ICH Q10 system demands two core ingredients:
- Controlled risk investigation and management
- Timely and appropriate quality responses to product quality feedback, such as parameter and attribute measurements, complaints, non-conformances, deviations, audit findings and so on
Apply the audit management functionality to dig into and interrogate the quality of your product and processes.
Build flexible and completely paperless digital audits to uncover gaps, risks, weaknesses and improvement opportunities, pushing your findings into reports instantly at the touch of a button. Schedule regular audits of your various departmental and operational areas, applying customized question banks and checklists to get the information you need to see.
Then push your uncovered risks, opportunities, complaints and non-conformance reports (NCRs) into Ideagen Quality Management’s issue management module for immediate contextual response.
Build bespoke workflows to handle any kind of business action, from squashing a non-conformance with a CAPA plan to responding to adverse events, systemic issues and customer feedback. Funnel actions to the right personnel at the right time for a consistent, snappy, targeted and fully audit-trailed response that maintains and improves the quality of your processes and final product.
Corrective and preventive action (CAPA) management
Closely overlapping with your ability to monitor and maintain your process integrity, the second pillar of your ICH Q10 system is your ability to determine the root cause of adverse events and to take structured and appropriate CAPA steps which continually improve product quality.
Ideagen Quality Management’s issue management module is designed to allow users to build closed-loop CAPA pathways.
Custom issue forms encourage all members of staff to be the eyes and ears of the quality department, raising and reporting issues quickly and easily and pushing them directly into the Ideagen Quality Management system for remediation.
Stepped workflows guide designated staff through the stages of each CAPA process, ensuring due diligence while minimizing human error or negligence. Best of all, every CAPA managed in Ideagen Quality Management is fully traceable, allowing you to prove to your auditors that the correct process was followed and appropriate action was taken.
Change management
Effective ICH Q10 change management means proposed changes are properly evaluated, executed and documented, minimizing adverse risk consequences while driving continuous improvement and maintaining compliance.
Through our solutions, you can create a living, breathing library of international legislative requirements.
Use the document management module to record, store and categorize any associated business records.
Use built-in version control to update policies, processes and procedures, confident that superseded and outdated documents are withdrawn from circulation. A single central business repository ensures all employees are connected and aware of changes as and when they happen – including acknowledging standard operating procedure (SOP) updates or understanding new processes.
Management review of process performance and product quality
Your senior management should provide the coordination, oversight and support to assure the end-to-end lifecycle quality of your products and processes. Ideagen Quality Management gives your business a centralized single source of truth. This means it’s easy for management to view the entire quality landscape of your pharmaceutical operation at a glance and drive your business in a unified direction.
Store quality manuals, statements, processes and policies in the document module to demonstrate management’s commitment to quality and set frequent review intervals to keep them relevant and up-to-date. Document your management review meetings, storing records in the document module and pushing agreed actions into the issues module for immediate follow-up.
Using the reporting functionality of the audit module provides instant summary reports of audits and inspections to management, highlighting risks and improvement opportunities for discussion and response.
As Ideagen Quality Management users, it’s easy for management to allocate and manage system resources – assigning key personnel for workflow steps, setting bespoke viewing permissions to share key documents with specific areas of the business and building and sharing reports to drive smart data-driven decision-making.
Any piece of data within the system can be interrogated and reviewed by management, including:
- Outstanding CAPA process steps
- Who has read and acknowledged an updated procedure and who hasn’t
- The number of findings per audit and per business area
With complete knowledge and visibility of a central real-time QMS, it’s simple for management to plan, cultivate and continually measure a top-down quality-centric approach that satisfies your customers and regulators.
Applying Ideagen quality solutions
Looking to build an ICH Q10 pharmaceutical quality system that’s centralized, digitized and data-driven? Ideagen Quality Management is a tool for all layers of your business, from lab staff to C-suite, to coordinate and continually improve your pharmaceutical quality.
Applying Ideagen’s quality software to meet the ICH Q10 guideline is a highly beneficial decision for any pharmaceutical organization. Ideagen's solutions provide a comprehensive and robust framework to manage GMP guidelines and regulations effectively.
With proven reliability across the global pharmaceutical industry, this software ensures adherence to unbreakable standards of quality, facilitates thorough documentation and tracking of compliance activities and supports continuous improvement initiatives. By leveraging Ideagen's advanced technology, organizations can streamline their quality processes, ensure regulatory compliance and ultimately enhance product quality and patient safety.
Aaron is an experienced content writer specializing in quality and compliance, with a focus on regulatory standards and enhancing operational practices. With a strong background in the life sciences and manufacturing industries, he excels at creating clear, impactful content that supports organizations in achieving sustainable excellence and meeting complex industry requirements.


